How Many Sig Figs In 100.0
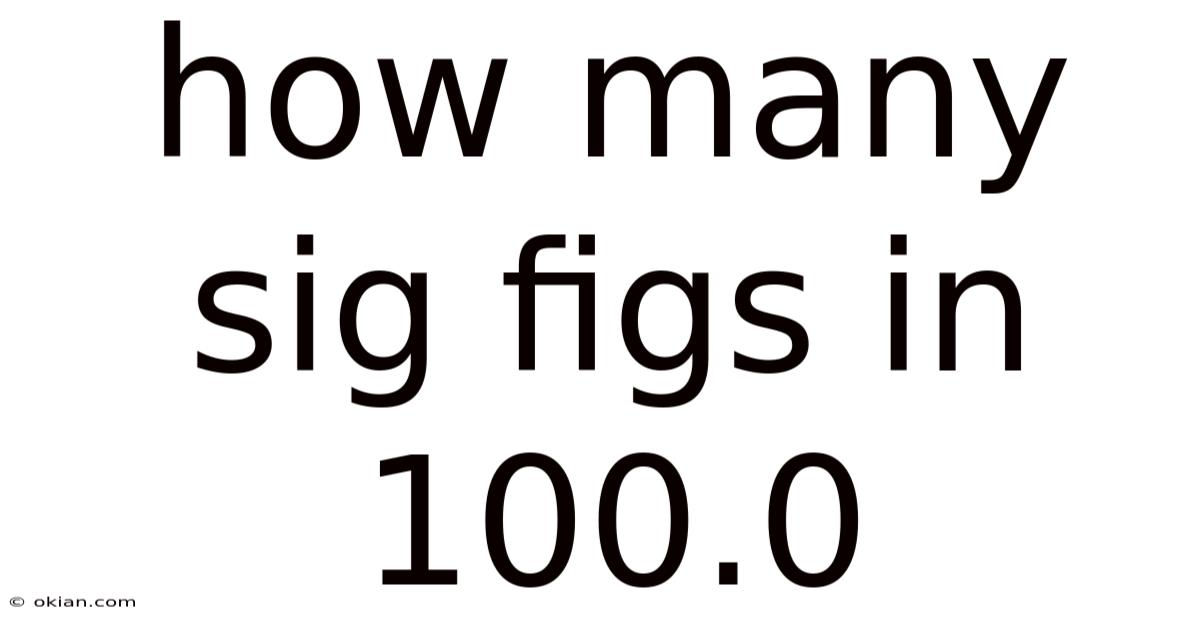
Understanding Significant Figures: Why 100.0 Has Four, Not Three
In the precise world of science, engineering, and data analysis, the way we write numbers is not just about quantity—it’s a direct statement about the precision and confidence we have in our measurements. A seemingly simple number like 100.0 holds a wealth of information for those who understand the rules of significant figures (often abbreviated as "sig figs"). Determining how many significant figures are in 100.0 is an excellent lesson in this critical language of accuracy. The short answer is that 100.0 contains four significant figures. This article will comprehensively explain why this is the case, exploring the foundational rules, common pitfalls, and the profound importance of this concept in real-world applications.
Detailed Explanation: The Grammar of Precision
Significant figures are the digits in a number that carry meaning contributing to its measurement resolution. They are essentially the "trustworthy" digits, excluding any that are merely placeholders. The entire system exists to prevent the false implication of precision. For example, if you measure a length with a ruler marked only in centimeters and get 10 cm, writing it as 10.00 cm would be dishonest—your tool couldn't justify that level of detail. Sig figs enforce honest communication of uncertainty.
The rules for identifying significant figures are a consistent set of guidelines:
- All non-zero digits are significant. (1, 2, 3, 4, 5, 6, 7, 8, 9 are always counted).
- Zeros between non-zero digits are significant. (e.g., in 101, the zero is significant).
- Leading zeros (zeros that precede all non-zero digits) are NOT significant. They only set the decimal place. (e.g., 0.005 has one sig fig; the zeros are placeholders).
- Trailing zeros (zeros that come after all non-zero digits) are significant ONLY if the number contains a decimal point. This is the crucial rule for our number, 100.0.
The presence or absence of a decimal point is the key that unlocks or locks the significance of trailing zeros. A decimal point signals, "We measured or calculated to this specific decimal place." Without it, trailing zeros are ambiguous—they could be significant or just placeholders. With it, they are definitively part of the measured value.
Step-by-Step Breakdown: Analyzing 100.0
Let's dissect the number 100.0 digit by digit using the rules:
- First Digit (1): This is a non-zero digit. It is significant. (Rule 1).
- Second Digit (0): This zero is between the non-zero digits '1' and the next non-zero digit? Not exactly. It is a trailing zero relative to the first '1', but we must look at the entire number. More importantly, it is a zero that is followed by a decimal point and another digit. The decimal point's presence is the deciding factor for all trailing zeros.
- Third Digit (0): This is another trailing zero. Again, its significance is determined by the decimal point.
- Fourth Digit (0): This is the digit immediately after the decimal point. Any digit placed after a decimal point is, by definition, a measured or specified digit. It is significant. (Rule 4, applied directly).
Because the number 100.0 explicitly contains a decimal point, all trailing zeros become significant. Therefore, we count:
- The '1' (1 sig fig)
- The first '0' (2nd sig fig)
- The second '0' (3rd sig fig)
- The '0' after the decimal (4th sig fig)
Total: Four significant figures.
The Critical Contrast: 100 vs. 100.0 vs. 100.00
This distinction becomes crystal clear when comparing:
- 100: Has one significant figure (only the '1'). The two zeros are trailing zeros without a decimal point, so they are ambiguous placeholders. It implies a measurement somewhere between 50 and 150.
- 100.0: Has four significant figures. The decimal point declares that the measurement was precise to the tenths place. It implies a measurement between 99.95 and 100.05.
- 100.00: Has five significant figures, indicating precision to the hundredths place.
Real-World Examples: Where This Distinction Matters
Imagine a chemist recording the mass of a compound.
- If the balance reads 100.0 grams, the chemist knows the mass is 100.0 ± 0.05 grams. The instrument's precision is to 0.1 grams. This level of detail is crucial for stoichiometric calculations in a reaction.
- If the same mass were recorded as 100 grams (from a less precise scale or by rounding), the implied uncertainty is ± 50 grams. Using this value in a chemical formula could lead to a dramatically wrong amount of reactant, potentially ruining an experiment or creating a safety hazard.
In engineering, specifying a shaft diameter as 100.0 mm versus 100 mm is a world of difference. The former suggests a machining tolerance on the order of ±0.05 mm, achievable with precision lathes.
In high-stakes industries like medical device manufacturing, such precision is non-negotiable. A catheter length specified as 100.0 mm conveys a tolerance of ±0.05 mm, a requirement for proper fit and function. Recording it as 100 mm would imply a much cruder tolerance of ±50 mm, rendering the part useless and potentially dangerous. This principle extends to financial modeling and data science as well, where the number of significant figures in input data dictates the meaningful precision of computational results and reported confidence intervals. Overstating precision (e.g., treating 100 as having three sig figs) creates a false sense of accuracy, while understating it (e.g., rounding 100.0 to 100) discards valuable information.
Ultimately, significant figures are not merely an academic rule but a universal language of uncertainty. The presence or absence of a decimal point acts as a critical switch, determining whether trailing zeros are meaningful data points or ambiguous placeholders. This distinction allows scientists, engineers, and technicians to implicitly communicate the reliability and resolution of their measurements. Recognizing and applying this rule ensures that numerical data is transmitted with integrity, preventing costly errors, unsafe designs, and flawed conclusions. In a world driven by quantitative analysis, the ability to read and write numbers with correct significance is a fundamental safeguard for both progress and safety.
Latest Posts
Latest Posts
-
Draw The Lewis Dot Structure For Co
Mar 28, 2026
-
How To Express F In Standard Form
Mar 28, 2026
-
What Was The Purpose Of The Grange Movement
Mar 28, 2026
-
Find The Area Of The Region That Is Not Shaded
Mar 28, 2026
-
Why Do Scientists Consider Vestigial Structures Evidence For Evolution
Mar 28, 2026