What Is The Mass Of 9.30 Moles Of Sih4
okian
Mar 11, 2026 · 5 min read
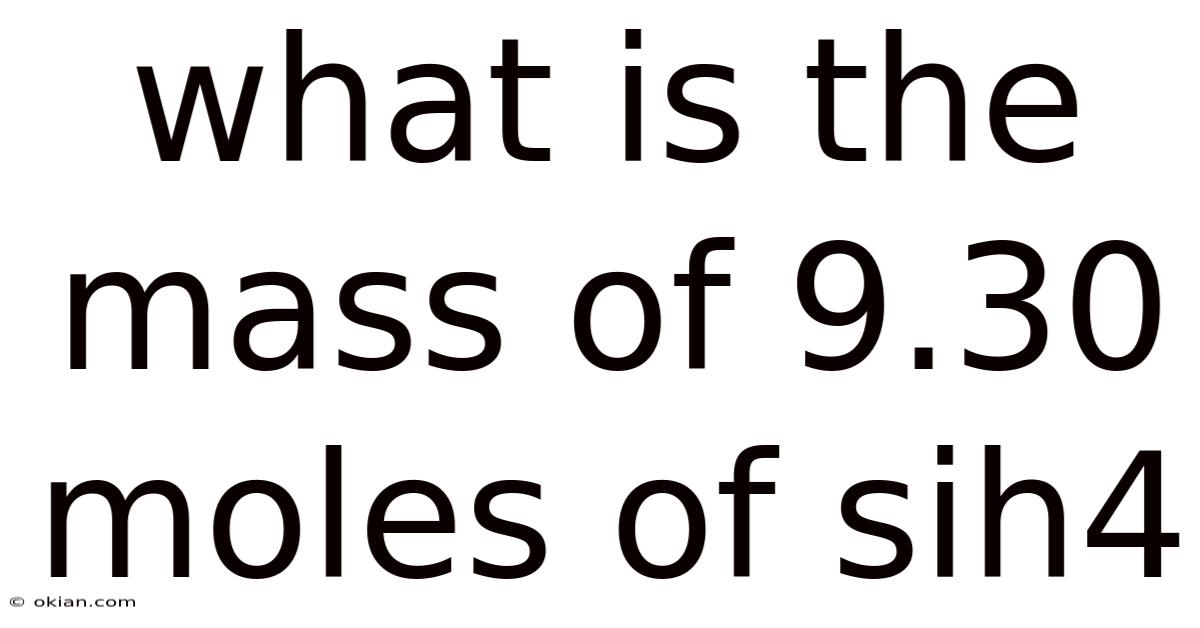
Table of Contents
What is the Mass of 9.30 Moles of SiH₄?
Introduction
The question what is the mass of 9.30 moles of SiH₄ is a fundamental calculation in chemistry that bridges the gap between the microscopic world of atoms and the macroscopic measurements we observe in laboratories or industrial settings. This calculation is not just a simple arithmetic problem; it represents a critical application of the mole concept, which is one of the cornerstones of modern chemistry. By understanding how to determine the mass of a given number of moles of a substance, students, researchers, and professionals can accurately predict the quantities of reactants and products in chemical reactions, design experiments, and ensure safety in industrial processes.
The term mass of 9.30 moles of SiH₄ refers to the total weight of 9.30 moles of silicon tetrahydride (SiH₄), a compound composed of one silicon atom and four hydrogen atoms. This calculation is essential because it allows chemists to convert between the amount of substance (measured in moles) and its mass (measured in grams), a conversion that is vital for stoichiometric calculations. For instance, if a reaction requires a specific amount of SiH₄, knowing its mass ensures that the correct quantity is used, preventing errors that could lead to failed experiments or hazardous situations.
This article will explore the concept of molar mass, break down the step-by-step process of calculating the mass of 9.30 moles of SiH₄, provide real-world examples of its application, and address common misconceptions. By the end, readers will have a clear understanding of why this calculation is significant and how it is applied in both academic and practical contexts.
Detailed Explanation
To fully grasp what is the mass of 9.30 moles of SiH₄, it is essential to understand the underlying principles of molar mass and the mole concept. Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). It is calculated by summing the atomic masses of all the atoms in a molecule. For SiH₄, this involves adding the atomic mass of silicon (Si) and four times the atomic mass of hydrogen (H). The atomic mass of silicon is approximately 28.085 g/mol, while hydrogen is about 1.008 g/mol. Multiplying
each atomic mass by the number of atoms in the molecule (4 for SiH₄) and then summing these values gives the molar mass of SiH₄.
So, for SiH₄:
- Atomic mass of Si = 28.085 g/mol
- Atomic mass of H = 1.008 g/mol
- Number of H atoms per molecule = 4
Molar mass of SiH₄ = (1 * 28.085 g/mol) + (4 * 1.008 g/mol) = 28.085 g/mol + 4.032 g/mol = 32.117 g/mol
Now that we have the molar mass of SiH₄, we can calculate the mass of 9.30 moles:
Mass = (Moles) * (Molar Mass) Mass = 9.30 moles * 32.117 g/mol Mass = 290.0 g
Therefore, the mass of 9.30 moles of SiH₄ is approximately 290.0 grams.
Real-World Applications
The calculation of the mass of 9.30 moles of SiH₄ isn't confined to the laboratory. It has wide-ranging applications across various fields. In the chemical industry, this calculation is crucial for optimizing industrial processes. For example, in the production of silicon-based materials, precise stoichiometric control is vital. Knowing the exact mass of reactants allows for efficient and cost-effective production.
Furthermore, in pharmaceutical research, accurately determining the mass of SiH₄ is important when synthesizing novel drug candidates. Incorrect mass calculations could lead to inaccurate yields and potentially unsafe products. The calculation also plays a role in environmental chemistry, where understanding the quantities of chemicals involved in industrial emissions is essential for monitoring and mitigating pollution.
Beyond these industrial applications, the concept is fundamental to chemical education. Students learn this calculation as a core skill for understanding chemical reactions, predicting outcomes, and designing experiments. It provides a tangible link between abstract chemical concepts and real-world applications, solidifying their understanding of stoichiometry.
Addressing Common Misconceptions
One common misconception is confusing the molar mass with the molecular weight. While they are related, molar mass is defined as the mass of one mole of a substance, while molecular weight is the mass of one molecule of a substance. For compounds, the molecular weight is the sum of the atomic weights of all atoms in the molecule. The molar mass is the mass of one mole, which is a different quantity. Another frequent mistake is neglecting to account for the number of atoms in a molecule. Simply adding the atomic masses of the constituent atoms doesn't accurately represent the molar mass; you must multiply by the number of atoms in the molecule.
Conclusion
In conclusion, calculating the mass of 9.30 moles of SiH₄ is a fundamental yet powerful application of the mole concept. It demonstrates the direct relationship between the amount of a substance (moles) and its mass (grams), a relationship essential for accurate stoichiometric calculations in chemistry. From industrial optimization and pharmaceutical research to educational purposes, this calculation has broad implications. By understanding molar mass and the proper application of these principles, chemists and students alike can confidently navigate the complexities of chemical reactions and contribute to advancements in various scientific and technological fields. The seemingly simple calculation of mass reveals the intricate and interconnected nature of the chemical world, highlighting the importance of precise quantitative analysis in understanding and manipulating matter.
Latest Posts
Latest Posts
-
What Are The Stages Of The Demographic Transition
Mar 11, 2026
-
Ap Psych Unit 5 Practice Test
Mar 11, 2026
-
What Is The Use Of A Capacitor In A Circuit
Mar 11, 2026
-
Real World Example Of Longitudinal Wave
Mar 11, 2026
-
Which Of The Following Is An Example Of Artificial Selection
Mar 11, 2026
Related Post
Thank you for visiting our website which covers about What Is The Mass Of 9.30 Moles Of Sih4 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.